Cell And Gene Therapy: Oncology Clinical Trial Trends In The United States
Cell and Gene Therapy: Oncology Clinical Trial Trends in the United States
- The key takeaways of cell and gene therapy
- Extensive background on the uptake of cell and gene therapy
- Insights and outlook of cell and gene therapy trials in the US
- An analysis of the top oncology cell and gene therapy assets and developers
Thank you.Please check your email to download the Whitepaper.
Although breast cancer remains a leading cause of cancer death in both developing and developed nations, World Health Organization statistics show that survival rates in richer countries often exceed 80%. This is primarily due to improvements in early screening, diagnosis and treatment over the past three decades.
Focusing on drugs, most of this success has been in the two most common types of breast cancer hormone receptor and human epidermal growth factor receptor 2 positive . HR+ breast cancers express the oestrogen receptor , sometimes as well as the progesterone receptor , and therefore grow in the presence of those hormones, while HER2+ cancers over-express the HER2 gene.
These two sub-types have seen the most progress as drug development from the 1970s onwards has focused on uncovering and resolving the hormonal basis of breast cancer with anti-oestrogen therapies. Moreover, pharma companies uncovered methods of targeting the HER2 oncogene, therefore inhibiting the growth of any tumours expressing that gene.
Where Do These Numbers Come From
The American Cancer Society relies on information from the Surveillance, Epidemiology, and End Results Program database, maintained by the National Cancer Institute , to provide survival statistics for different types of cancer.
The SEER database tracks 5-year relative survival rates for breast cancer in the United States, based on how far the cancer has spread. The SEER database, however, does not group cancers by AJCC TNM stages . Instead, it groups cancers into localized, regional, and distant stages:
- Localized: There is no sign that the cancer has spread outside of the breast.
- Regional: The cancer has spread outside the breast to nearby structures or lymph nodes.
- Distant: The cancer has spread to distant parts of the body such as the lungs, liver, or bones.
What Is The Treatment For Triple
Chemotherapy.Chemotherapy is almost always called for, Sun says. Chemo can downstage tumors . While Sun says the chemotherapy for triple-negative breast cancer can be intense, she adds that regimen can be tailored to the individual and adjusted for older or frailer patients.
In those cases where we get complete response, we know we gave you the right medicine and your prognosis is good, Sun says.
Surgery can remove more of the tumor. Surgery for triple-negative breast cancer does not always have to be a mastectomy, Sun says. Effective chemotherapy done first opens up the possibility of less-invasive surgical options that are less of an ordeal for the patient. If the tumor is small enough after chemo, outpatient procedures or a lumpectomy may be possible.
Surgical samples of the cancerous tissues taken from surgery can provide more information on the cancer and how it is behaving so chemotherapy can be tailored accordingly.
Radiation therapy involves the use beams of radiation to destroy cancer cells, using various techniques to prevent damage to healthy surrounding tissue.
Medical treatments are being tested on triple-negative breast tumors in clinical trials.
Immunotherapy and PARP inhibitors are very exciting and theres lots of research going on, including here at Johns Hopkins, Sun says.
Read Also: Can Chemotherapy Cure Breast Cancer
Next Step: Multinational Phase Iii Trials
Any side effects from the treatment in each case a result of the docetaxel, not the L-NMMA were minor and quickly resolved, according to Dr. Chang.
L-NMMA was initially developed for the management of cardiogenic shock a phase III trial investigating such use found it was safe but of no benefit before Dr. Chang’s team tried it on cancer. She made a deal to secure the drug from a Cornell University pharmacologist who led the trial and still had vast supplies.
The next step for Dr. Chang and the team are multinational phase III trials to evaluate the L-NMMA and chemotherapy combination in a significantly larger number of TNBC patients. If those trials show positive results, the team plans to file for FDA approval of L-NMMA.
In addition, Houston Methodist plans to seek an orphan indication from the FDA for metaplastic breast cancer, an aggressive subtype of TNBC that accounts for less than 1% of all breast cancers. The National Cancer Institute has awarded Houston Methodist a grant to take the therapy into global phase III clinical trials, which will begin in late 2022.
Dr. Chang also said she plans to research the combination of L-NMMA and checkpoint blockade immunotherapy against triple negative breast cancer. Such immunotherapy has revolutionized treatment of many cancers, but by itself has only shown modest benefits against TNBC.
Taking Care Of Yourself

After your treatment is over, your doctor will want to see you often to make sure the cancer doesn’t return. For the first 3 years, you’ll likely see them every 3 to 6 months. For 2 years after that, you’ll probably visit every 6 to 12 months. Once you’ve been cancer-free for 6 years, you’ll probably go back only once a year. Tell the doctor right away if you get any new symptoms or if you have pain or other problems that relate to your breasts.
Also Check: Mortality Rates Of Breast Cancer
Utilising New Vaccine Technology
Research is all about finding new and innovative ways to treat breast cancer. This comes down to better understanding how different processes in the body, that are linked to breast cancer, work and how different molecules can be targeted to provide new treatments for the disease.
One area of research with amazing recent breakthrough is mRNA vaccine technology. You may have heard of this technology, as this was used to develop the Pfizer-BioNTech COVID-19 vaccine.
The vaccine technology works by presenting instructions to our immune cells so they can recognise certain molecules. For example, this could be a molecule found on the outside of a cancer cell. These instructions help the immune system to recognise the cancer cell, and kill it more quickly.
Scientists hope that this technology can be used to create new treatments for certain cancers, like triple negative breast cancer.
Research Progress And Future Questions
After the 2020 approval of the combination of pembrolizumab and chemotherapy for advanced triple-negative breast cancer, FDA approved the combination therapy for people with early-stage disease in 2021.
That approval was based on results from a different trial, KEYNOTE-522. In that study, patients with high-risk, early-stage triple-negative breast cancer benefited from pembrolizumab given with chemotherapy before surgery, and then continued as a single agent as an additional, or adjuvant, treatment after surgery.
This is an exciting time for research on triple-negative breast cancer, said Dr. Lee. We have now seen a benefit from an immune checkpoint inhibitor and chemotherapy in a subgroup of patients in both the advanced and early stages of the disease.
Dr. Lee cautioned, however, that more than half of all patients with triple-negative breast cancer have PD-L1 combined positive scores of less than 10, so more work is needed to find effective treatments for these patients.
In his editorial, Dr. Pivot noted that people diagnosed with triple-negative breast cancer are not a homogeneous group. Future studies, he added, will try to identify which individuals are more or less likely to benefit from pembrolizumab.
Also Check: Can Guys Get Breast Cancer
Clinical Trial Is Logical Next Step
And so when we come across a drug that potentially could work, especially in this population that has a high prevalence of EGFR mutation, something we can capitalize on to treat this patient population, Dr. Pathak added.
However, she stressed this is still in the early stages and that testing in human patients still needs to be done:
I think the logical next step is to see how it works in humans phase 1 trials seeing is it tolerable? And then additionally, it would be interesting to see how we can use this drug in other ways in terms of cancer therapy. We target the growth mechanism of the cancer, stopping its growth. But another aspect is making sure we can stop its spread the spread of it is a different mechanism. So to see if you could stop both types of issues within this presentation.
Update On Strategies To Improve Treatment For Triple Negative Breast Cancer
Approximately 12% of all breast cancers are TNBC, meaning that they are estrogen-receptor negative , progesterone-receptor negative , and human epidermal growth factor receptor 2-negative . This means that TNBC is not stimulated to grow from exposure to the female hormones estrogen or progesterone, nor through an overactive HER2 pathway.
Historically, most available and effective treatment options for the majority of breast cancers block the growth stimulating effects of ER, PR and/or HER2 therefore, TNBC has had limited therapeutic options.
In addition, TNBC tends to be an aggressive type of cancer, is often diagnosed at a more advanced stage, and affects younger women more often than other breast cancers. Novel treatment options for TNBC have lagged behind that of other types of breast cancers but the treatment landscape is improving.
When patients are newly diagnosed with TNBC, they should undergo NGS-biomarker testing to check for PD-L1 status, BRCA and other biomarkers that can identify individuals who can benefit from treatment with newer precision cancer medicines and immunotherapy.
Also Check: How Many Women Have Breast Cancer
Overall Advances In Breast Cancer Treatment
The prognosis for breast cancer patients has improved significantly over the decades, which is thought to be due to improved early diagnosis methods and the provision of post- or pre-operative adjuvant therapy. According to Wiklund, the June conference also showed for the first time that the average overall survival rate of all breast cancer patients has also improved thanks to the development of drug therapy for advanced breast cancer.It is significant that, in the treatment of advanced breast cancer, we have an increasing range of effective drugs at our disposal to significantly prolong the life expectancy of severely ill patients. There have been major breakthroughs in cancer treatment in recent years. Metastatic breast cancer can nowadays often be treated as a chronic disease. At Docrates, we are already applying these newest treatments actively, says Tom Wiklund.
Tom Wiklund, an Associate Professor in Oncology, is a Specialist in Oncology at Docrates Cancer Center. He has specialised in the treatment of breast cancer, testicular cancer, sarcomas and lymphomas, as well as in the long-term follow-up of cancer patients and the late adverse effects of treatment. Wiklund has extensive experience in clinical work, international cooperation and clinical trials. He has also taken an active part in many oncological working groups in the Nordic countries and collaborated with cancer registries in Finland and the United States.
Improving Treatment For Triple
A team led by Northwestern Medicine investigators has identified a novel molecular target that may improve the efficacy of current treatments for patients with triple-negative breast cancer, according to findings in the journal EMBO Molecular Medicine.
This study demonstrates an alternative way to treat this larger subset of patients with triple-negative breast cancer, said Yong Wan, PhD, professor of Obstetrics and Gynecology in the Division of Reproductive Science in Medicine, of Pharmacology and senior author of the study.
Massimo Cristofanilli, MD, professor of Medicine in the Division of Hematology and Oncology, was a co-author. Zhuan Zhou, PhD, a former postdoctoral fellow in the Wan laboratory, was the lead author of the study.
Triple-negative breast cancer cells do not display any of the hormone receptors more commonly targeted in breast cancer: estrogen, progesterone or the HER2 protein. The absence of these receptors therefore allows the cancer cells to be insensitive to endocrine therapies, or hormonal thearpies, and proliferate relatively faster than other types of breast cancers.
However, only 20 percent of patients with TNBC test positive for the BRCA mutation and respond successfully to PARP inhibitors, leaving the remaining 80 percent of patients with very limited and harsh treatment options.
This work fills a molecular gap for KLF4 in regulating DNA damage repair, Wan said.
You May Like: How To Look For Breast Cancer
New Approach For Triple Negative Breast Cancer
About 10-15% of breast cancer cases are diagnosed as triple negative breast cancer, which means the cancer cells lack all three receptors that doctors can target with cancer-treating medications. As there are currently no effective targeted therapies, triple negative breast cancers have a poorer prognosis compared to other forms of breast cancer.
Research from Associate Professor Chaffers team revealed that triple negative cancer cells switch their cell state in response to chemotherapy, which not only makes the cancer cells more aggressive, but also allows them to evade treatment.
We found that chemotherapy triggers a cell change in cancer cells that enables them to build a defence against the chemotherapy. This means that a different type of cancer cell emerges after treatment, which has become resistant to the chemotherapy and is a major cause of cancer relapse, Associate Professor Chaffer explains.
We aim to put a stop to this cancer resistance strategy to improve the effectiveness of chemotherapy for triple negative breast tumours.
Dr Beatriz Perez San Juan, the post-doctoral researcher in Associate Professor Chaffers lab who led the preclinical study, discovered that activation of androgen receptors in breast cancer cells triggers the cell state switching. Androgens are commonly thought of as male sex hormones but are also found at lower levels in women.
New Breakthrough May Advance Triple Negative Breast Cancer Treatment
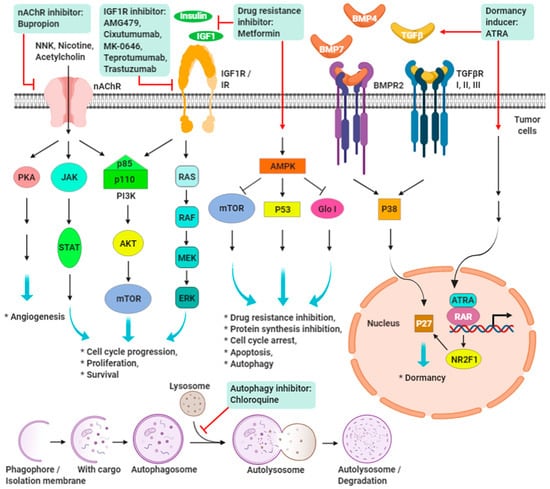
Scientists at The Scripps Research Institute have developed a new drug candidate that lowers the growth rate of tumor cells in animal models1 in one of the most difficult cancers to battle: triple negative breast cancer. This is the first time that scientists have been able to take a genetic sequence and design a drug candidate that is effective against this type of cancer. What makes the molecule so unique is its ability to destroy only the cancer cells that express the cancer gene, leaving healthy cells intact. The research is poised to totally change the way we identify drugs: by working with the genetic makeup of an illness.
The compound designed by the scientists is called Targaprimir-96 it causes breast cancer cells to destroy themselves by targeting a specific RNA that sparks the cancer precisely. In the past, scientists were forced to test millions of possible drug candidates to find a few that successfully affected target areas. The new study makes the process much speedier and more effective. It relies on an approach called Inforna, which is centered on creating compounds that bind to RNA folds. They are particularly effective at targeting microRNAs.
References
Read Also: What Are The Most Common Types Of Breast Cancer
Clinical Trials For New Cancer Vaccine Could Start Within Three Years
Scientists, funded by Worldwide Cancer Research, are ready to start clinical trials for a new cancer vaccine thanks to a recent research breakthrough. The team, led by Associate Professor Kristen Radford in Queensland, Australia, hope that they will be able to begin clinical trials within the next three years and believe that their findings will be relevant for many different types of cancer, including breast cancer.
Repurposing An Experimental Treatment
Seviteronel was originally developed as a standalone therapy for breast and prostate cancers that carry the androgen receptor and was proven safe for patients in Phase II clinical trials.
Our research has revealed that seviteronel may be far more beneficial as an adjunct therapy. We found that androgen inhibition blocks cancer cell state switching, locking cancer cells in a chemotherapy-sensitive state. This is why chemotherapy plus seviteronel treatment was more effective than chemotherapy alone at targeting cancer in our preclinical studies, says Dr Perez San Juan.
We hope that this new combination treatment approach will drastically reduce drug resistance to improve the effectiveness of standard-of-care chemotherapy and, ultimately, improve outcomes for patients.
For further information about the 4CAST clinical trial or to register your interest for the trial, please contact St Vincents Hospital Sydney Research Office,
–ENDS
The clinical trial is sponsored by St Vincents Hospital Sydney and supported by Kembi Therapeutics Pty Ltd. The trial was made possible by research supported by The NELUNE Foundation, The Paramor Family, Australias National Health & Medical Research Council, Cancer Institute NSW, the National Breast Cancer Foundation, the Girgensohn Foundation and the St Vincents Hospital Research Foundation.
The research has ethics approval from St Vincents Hospital Human Research Ethics Committee .
Read Also: Can You Lose Hair From Breast Cancer
Combinations Of Immune Checkpoint Inhibitors
However, most patients with TNBC do not respond well to PD-1 or PD-L1 monotherapy therefore, inducing a favorable tumor immune microenvironment appears to be particularly important. Conventional chemotherapeutic agents, such as taxane, cisplatin, and cyclophosphamide, can enhance tumor antigen release, improve the tumor microenvironment, and add the possibility of an antitumor response . Biopsies before and after NAC showed that the immune microenvironment was altered from low TIL to high TIL, and patients with high TIL levels exhibited improved survival . For example, paclitaxel has pleiotropic immune-modulating effects because it helps mature dendritic cells shift the T-helper phenotype to promote the secretion of proinflammatory cytokines and enhance the activity of CD8+ T cells . An animal model has shown evidence that cisplatin markedly induces tumor regression and improves survival when combined with anti-PD-1 and anti-cytotoxic lymphocyte antigen 4 . These studies suggest that ICB combined with chemotherapy may achieve a synergistic or additive clinical effect.
In addition to the combination of PARPi and immunotherapy, a series of trials of immunotherapy in combination with other drugs have entered clinical studies . For example, alternative PD-1/PD-L1 ICB combined with tyrosinase inhibitors has entered phase III studies .
Table 4 Unpublished phase III trials of immunotherapy for TNBC