Study: New Genes Linked With Triple
A research study identified new genes that may be associated with a higher risk for triple-negative breast cancer.
This image is an example of the kind of gel pictures researchers use to understand the order, or sequence, of genes or to look for mutations in genes.
This image is an example of the kind of gel pictures researchers use to understand the order, or sequence, of genes or to look for mutations in genes.
Out of 100 people with breast cancer about 15 will have triple-negative breast cancer . Its one of the most challenging types of breast cancer to treat due to several things:
- TNBC tends to be more aggressive than other common types of breast cancer.
- Women usually dont learn they have it until the cancer has already spread to other parts of the body .
- Treatment options remain limited. Hormone therapies and targeted therapies that help treat people with some types of breast cancer, typically dont work for a woman with TNBC. Doctors use chemotherapy to treat these patients, but it isnt very effective and may have side effects.
If doctors had a way to identify women at risk for TNBC, they might be able to find cancer earlier and before it spreads when it might be easier to treat. And, if doctors had a way to personalize treatment for women with TNBC, they might be able to improve a womans outlook . One approach researchers are using to identify women at risk for TNBC and possible treatment options for TNBC is by studying inherited changes in cancer genes.
Other Cancer Risks For People With Hboc
Anyone with mutations in the BRCA2 gene may be at an increased risk of other types of cancer, including melanoma and pancreatic, stomach, esophageal, and bile duct cancers.Mutations in other genes may be associated with an increased risk of developing breast and other cancers, including the Li-Fraumeni syndrome , Cowden syndrome, and others. The pattern of cancers in the family is often a clue to the specific gene that may explain the hereditary cancer for that family. Multigene panels are available for people with a strong personal and family history of cancer. Multigene panel tests includeBRCA1 and BRCA2 and many other genes that increase the risk of breast, ovarian, and other cancers. If your BRCA1 and BRCA2 test was negative, then you may or may not have mutations in other genes. A newer type of testing, called next generation sequencing, massively parallel sequencing, or deep sequencing, has made testing for multiple genes at the same time faster and less expensive. If a genetic mutation is found, this could explain the cancers in a specific family and provide information about who is at risk and the appropriate types of monitoring and prevention/risk reduction methods.
Why Is Her2 Testing Important
Itâs important to know your HER2 status because doctors often recommend different therapies for HER2-positive breast cancer and HER2-negative breast cancer.
For instance, doctors treat many HER2-positive cancers with the medicine trastuzumab , which attaches to extra HER2 proteins and stops cancer growth.
Experts recommend that all people with invasive breast cancer be tested for HER2. Routine testing usually isnât done for a noninvasive type of breast cancer called ductal carcinoma in situ.
If your breast cancer returns or spreads, your doctor may retest the cells for HER2 abnormalities because results can change.
If you have breast cancer and donât know your HER2 status, talk to your doctor.
Show Sources
Read Also: What Happens In Breast Cancer
What Do Your Results Mean
Breast cancers with high levels of the HER2 protein are HER2-positive, and those with low levels of the HER2 protein are HER2-negative.
This generally means:
- If your IHC score is 0 to 1+, your cancer is HER2-negative.
- If your IHC score is 3+, your cancer is HER2-positive.
- If your IHC score is 2+, your cancer is âborderline.â Your doctor will probably recommend that you have the FISH test.
- If your FISH score is positive, your cancer is HER2-positive.
Whatâs tricky is that some breast cancers labeled HER2-positive can become HER2-negative over time. And HER2-negative breast cancer can switch to HER2-positive.
Researchers have found that sometimes HER2 test results are wrong. This can happen because pathologists who examine the samples use different lab processes to classify the tissue. Or tissue from one area of the breast may test positive, while tissue from another area can test negative.
You may want to talk to your doctor about the accuracy of your test results. If theyâre borderline, your doctor might recommend another test.
What Happens During A Her2 Breast Cancer Test
Most HER2 testing involves taking a sample of tumor tissue in a procedure called a biopsy. There are three main types of biopsy procedures:
- Fine needle aspiration biopsy, which uses a very thin needle to remove a sample of breast cells or fluid
- Core needle biopsy, which uses a larger needle to remove a sample
- Surgical biopsy, which removes a sample in a minor, outpatient procedure
Fine needle aspiration and core needle biopsies usually include the following steps:
- You will lay on your side or sit on an exam table.
- A health care provider will clean the biopsy site and inject it with an anesthetic so you won’t feel any pain during the procedure.
- Once the area is numb, the provider will insert either a fine aspiration needle or core biopsy needle into the biopsy site and remove a sample of tissue or fluid.
- You may feel a little pressure when the sample is withdrawn.
- Pressure will be applied to the biopsy site until the bleeding stops.
- Your provider will apply a sterile bandage at the biopsy site.
In a surgical biopsy, a surgeon will make a small cut in your skin to remove all or part of a breast lump. A surgical biopsy is sometimes done if the lump can’t be reached with a needle biopsy. Surgical biopsies usually include the following steps.
Don’t Miss: How Can You Check For Breast Cancer
Survival Rates And Statistics
A relative survival rate helps give an idea of how long a person with a particular condition will live after receiving a diagnosis compared with those without the condition.
For example, if the 5-year relative survival rate is 70%, it means that a person with the condition is 70% as likely to live for 5 years as someone without the condition.
It is important to remember that these figures are estimates. A person can talk with a doctor about how their condition is likely to affect them.
Some factors affecting a personâs survival rate with breast cancer include:
- individual factors, such as the personâs age and overall health
- the stage of the cancer at diagnosis
- the treatment the person receives
HER2-positive cancers are than HER2-negative cancers. With treatment, however, the chances of survival are high, especially with an early diagnosis. In some cases, they may be higher than for HER2-negative breast cancer due to effective targeted treatment.
According to the , the likelihood of living for another 5 years with HER2-positive cancer, compared with a person who does not have breast cancer, is as follows. These statistics are based on figures for the years 2011â2017.
| Stage |
|---|
Dont Miss: Can Stage 1 Breast Cancer Be Cured
What Causes Triple Negative Breast Cancer
Many patients wonder what causes triple negative breast cancer. The breast cancer experts at Moffitt Cancer Center are often asked this question, but as of yet, there are no clear answers. As research continues, more is being learned about the causes of breast cancer in general, and triple negative breast cancer specifically.
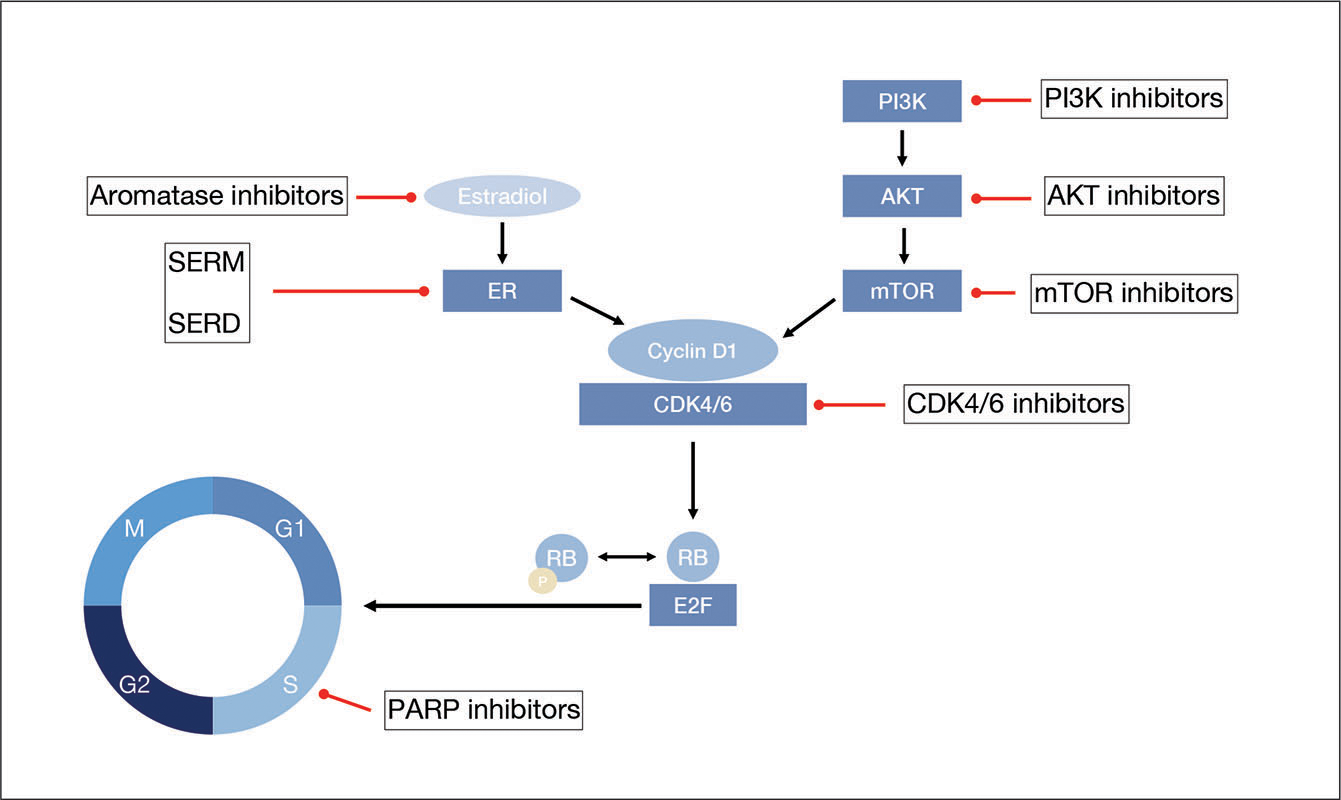
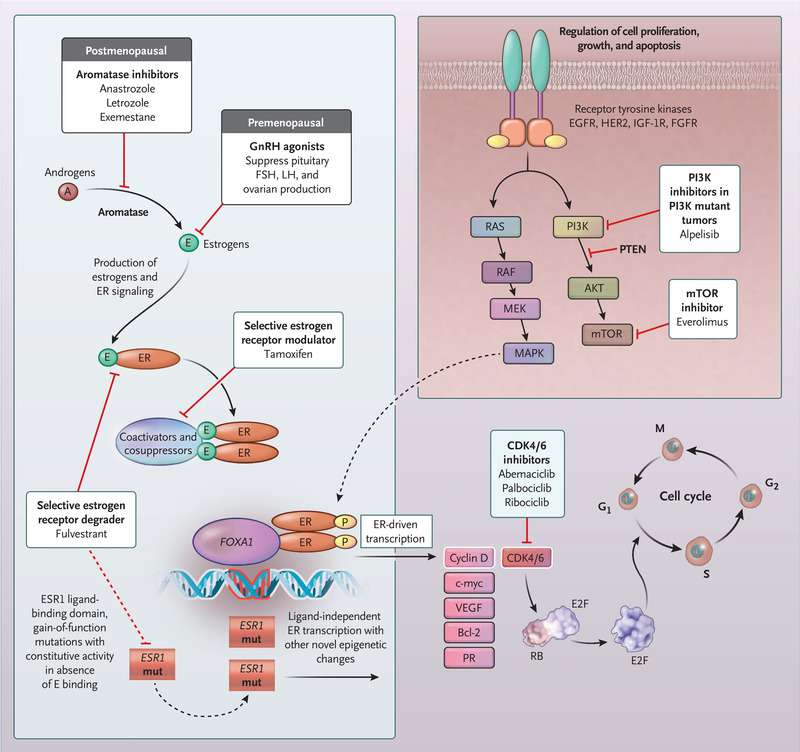
Triple negative breast cancer differs from other types of breast cancer in that the cancer cells do not have receptors for estrogen, progesterone or HER-2/neu hormones. When these receptors are present and exposed to the corresponding hormones, they can stimulate the cancer to grow. But, this also means that triple negative breast cancer patients do not benefit from hormone-based treatments, such as tamoxifen and Herceptin, which are sometimes effective for treating hormone-receptor-positive cancers.
The current theories on what causes triple negative breast cancer and hormone-receptor-positive cancers also differ. For instance, some breast cancers that grow in response to hormone exposure have been linked to a womans childbearing characteristics, such as the age at which she gave birth for the first time. Triple negative breast cancer does not seem to share this link.
Some researchers believe that one possible cause of triple negative breast cancer is a faulty BRCA1 gene. Here are some of the factors that support this theory:
- BROWSE
Also Check: Can Breast Cancer Be Cured
Sensitivity To Dual Her2 Blockade
Nowadays, an area with great interest for the oncologist community is to identify what patients might be treated with a regimen based on dual HER2 blockade without chemotherapy. It has been presented results of several neoadjuvant studies, which submit that a subgroup of patients with HER2+ BC are especially sensitive to the dual HER2 blockade, achieves pCR rates around 70%, so that could potentially be treated without chemotherapy .
The HER2-E breast tumors are driven by HER2/EGFR signaling, such as it showed, through a silico and omyc analyses, in the TCGA breast cancer project . So, this intrinsic subtype should benefit the most from anti-HER2 dual-blockade. The benefit achieved in HER-negative BC with HER2-E intrinsic subtype can be explained because these tumors preserve the higher expression of EGFR, with independence of expression degree of hormonal receptors . However, the greater response rate in the HER2-E subtype in previous studies could not distinguish anti-HER2 sensitivity vs. cytotoxic therapy-sensitivity. HER2-E subtype could be a predictor itself of anti-HER2 therapy benefit, and this theory should be validated in future randomized trials. If this happened, this intrinsic subtype could help to select a group of patients with HER2+ BC that might be cured with anti-HER2 drugs without chemotherapy, or patients with metastatic disease that can be treated with less intensive treatment, such as dual HER2 blockade-only.
Understanding Breast Cancer Risk Genes
A team of researchers in Rochester, Minnesota recently discovered mutations in 5 genes that seem to increase the risk for TNBC. These genes are known by several names: breast-cancer-risk genes, cancer-predisposition genes, and cancer-susceptibility genes. The research team was led by Fergus J. Couch, PhD, the Zbigniew and Anna M. Scheller Professor of Medical Research and Chair of the Division of Experimental Pathology and Laboratory Medicine at the Mayo Clinic.
Don’t Miss: Does Stage 1 Breast Cancer Require Chemotherapy
Breast Cancer Subtypes And Inherited Mutations
Cancers diagnosed in people with BRCA mutations often have specific characteristics:
-
60% to 80% of the breast cancers in women with a BRCA1 mutation are triple negative, as discussed above.
-
70% to 80% of the breast cancers in women with a BRCA2 mutation are estrogen receptor-positive, progesterone receptor-positive, and HER2-negative.
Her2 In Gastric Cancer
HER2 overexpression in patients with gastric cancer has been reported from 10 to 30% and correlates with poor outcome and a more aggressive disease. Overexpression of HER2 protein in gastric cancer, using immunohistochemistry , was first described in 1986 . In a study by Yano et al. , HER2 overexpression by IHC was found in 23% and gene amplification by FISH in 27% of 200 resected tumors. Gravalos and Jimeno in their study of 166 gastric cancer patients observed that HER2 overexpression was most commonly found in gastroesophageal junction tumors and tumors having intestinal type histology. Other studies also confirmed a higher rate of HER2 positivity in GEJ tumors and intestinal subtype . HER2 overexpression is directly correlated with poorer outcome in gastric cancer. In a study of 260 gastric cancers, HER2 overexpression was an independent negative prognostic factor and HER2 staining intensity was correlated with tumor size, serosal invasion, and lymph node metastases . Other studies also confirmed the negative impact of HER2 overexpression in gastric cancer .
Don’t Miss: How To Avoid Getting Breast Cancer
Reducing Your Risk Of Triple
Its difficult to reduce your risk of triple-negative breast cancer because you cant control your age or your race. However, women who test positive for the BRCA1 or BRCA2 gene mutations may want to consider preventive mastectomy. It can reduce the risk of breast cancer in these women by about 97%. But it isnt an easy decision. There are many things to consider before moving forward with surgery. During genetic testing for breast cancer, you will explore the risks and benefits to help you decide if its right for you.
Read Also: Does Breast Cancer Show Up In Blood Tests
Causes Of Triple Negative Breast Cancer
The risk factors for triple negative breast cancer are not clear. Some breast cancers depend on hormones to grow. These can be linked with risk factors to do with hormones and having children. But triple negative breast cancer does not seem to share these risk factors.
Most women with triple negative breast cancer have no strong history of breast cancer in their family . But some women with triple negative breast cancer have an altered BRCA1 gene. This will have been inherited from a parent.
An altered BRCA 1 gene can cause breast cancer to run in families. Most breast cancers caused by BRCA1 are triple negative.
If you have triple negative breast cancer, you may be offered genetic testing. This is even if you do not have a family history of breast cancer. Your cancer doctor or breast care nurse can explain more about this to you.
The tests are the same as for any type of breast cancer. You usually have a:
- Mammogram
A mammogram is an x-ray of the breast.
- Ultrasound scan
An ultrasound scan uses sound waves to produce a picture of the breast tissue and the lymph nodes in the armpit.
- Breast biopsy
When you have a breast biopsy, your cancer doctor or breast care nurse takes small samples of cells or tissue from your breast. The samples are looked at under a microscope to check for cancer cells. They also do other tests to find out if the cells have receptors for hormones, or for HER2.
See also
Read Also: What Leads To Breast Cancer
Common Susceptibility Loci For Brca1 Mutation Carriers
Because TNBC and breast cancer in BRCA1 mutation carriers are phenotypically similar, studies of genetic modifiers of breast cancer risk in BRCA1 mutation carriers have provided further insight into the genetic risk factors for TNBC. Specifically, SNPs in the 19p13.1 locus that displayed genome-wide significant associations with breast cancer in a GWAS of BRCA1 mutation carriers have also been associated with TNBC risk in the general population . In BCAC and TNBCC combined, the 19p13.1 locus was associated with TNBC risk but was not associated with the risk of ER-positive or ER-negative nontriple-negative breast cancer . Furthermore, these variants seemed to be specifically associated with tumors that were positive for the basal markers CK5/6 or EGFR , indicating specificity for the basal subtype. In addition, variants in ESR1, PTHLH, TOX3, CASP8, and TERT are also associated with both TNBC and the risk of breast cancer in BRCA1 carriers . Of the known TNBC risk factors, only RAD51L1 was not found to be a modifier of BRCA1-related breast cancer risk . Recent data also show that BRCA2 ER-negative tumors have pathologic characteristics similar to BRCA1 ER-negative tumors . Thus, further studies of BRCA1 and BRCA2 breast tumors, stratified by ER or triple-negative tumor status, may provide additional valuable insight into genetic susceptibility to TNBC.
Recommended Reading: Can I Survive Breast Cancer
Contribution Of Recently Identified Susceptibility Loci To Frr
The relative risk to daughters of an affected individual attributable to a given single nucleotide polymorphism was calculated using the formula :
Where p is the population frequency of the minor allele, q = 1 – p, and r1 and r2 are the subtype-specific genotypic relative risks ) for heterozygotes and rare homozygotes, relative to common homozygotes. For these calculations concordance in tumour type between proband and their relatives is assumed.
The proportion of the familial risk attributable to the SNP was then calculated as where 0 is the FRR for breast cancer to first degree relatives of cancer cases with each subtype, as estimated from our study. This formula assumes that the SNP of interest and other susceptibility alleles act multiplicatively on risk. Estimates of r1 and r2 and minor allele frequency for populations of European ancestry for each susceptibility allele were derived from the literature. Only SNPs that had been genotyped in breast cancer cases from Caucasian populations were included in the analysis.
Read Also: How To Treat Breast Cancer Without Chemotherapy
Her2 Mutation Status In Japanese Patients With Her2
We analyzed 286 HER2-negative breast cancers including 13 invasive lobular carcinomas. Among the 286 patients, two patients were found to have HER2 mutations, I767M and D769Y . Reverse sequencing also confirmed these results. In the case of the D769Y mutation, we also extracted genomic DNA from the patient’s blood. This DNA was distinct from the tumor DNA on the basis of the patient’s germline and somatic genotypes, indicating that the mutation was somatic. Both mutations were in the kinase domain. Moreover, both tumors were ER-positive invasive ductal carcinoma, not invasive lobular carcinoma .
The D769Y mutation in the kinase domain. Forward sequencing was performed. Reverse sequencing was performed and the mutation confirmed. Genomic DNA extracted from blood showed a wild-type sequence for the HER2 gene.
Asco Releases Rapid Guideline Recommendation Update For Patients With High
A new ASCO guideline update recommends offering 1 year of adjuvant olaparib to patients with high-risk, early-stage, HER2-negative breast cancer and germline BRCA mutations after completion of adjuvant chemotherapy and local treatment, including radiation. The recommendations update the 2020 guideline from ASCO, the American Society for Radiation Oncology , and the Society of Surgical Oncology published on the management of hereditary breast cancer.
The updated recommendation is the result of a new ASCO initiative aimed at providing clinicians with timely and practical guidance that reflects the latest practice-changing research. This recommendation update incorporates evidence from the OlympiA randomized controlled trial, following presentation at the 2021 ASCO Annual Meeting and publication in The New England Journal of Medicine.
Clifford A. Hudis, MD, FACP, FASCO
Guideline updates have traditionally required months to review new data, assemble an expert panel, develop, approve, and publish recommendationsmaking it difficult to provide timely support to clinicians seeking to keep abreast of rapidly advancing research. ASCOs new rapid guideline updating strategy is meant to address this need by providing rapid evidence review and dissemination of practice-changing trial results, said ASCO Chief Executive Officer Clifford A. Hudis, MD, FACP, FASCO.
More on OlympiA
2021 Updated Recommendation
Recommended Reading: When Was The First Case Of Breast Cancer Diagnosed