Region Of Interest Selection
Image selection
Two radiologists , who were blinded to the patients pathologic information, retrospectively reviewed the MRI images electronically on the picture archiving and communication system . In the case of disagreements, the two radiologists would reach a final decision by discussion and consensus. The final MRI images selected were exported from the PACS workstation in BMP format for histogram analysis. During export, the window width and window level were adjusted thus, all images had consistent window width and window level, and they were saved on the hard disk.
Histogram analysis
Figure 2Figure 3
N Categories For Breast Cancer
N followed by a number from 0 to 3 indicates whether the cancer has spread to lymph nodes near the breast and, if so, how many lymph nodes are involved.
Lymph node staging for breast cancer is based on how the nodes look under the microscope, and has changed as technology has gotten better. Newer methods have made it possible to find smaller and smaller groups of cancer cells, but experts haven’t been sure how much these tiny deposits of cancer cells influence outlook.
Its not yet clear how much cancer in the lymph node is needed to see a change in outlook or treatment. This is still being studied, but for now, a deposit of cancer cells must contain at least 200 cells or be at least 0.2 mm across for it to change the N stage. An area of cancer spread that is smaller than 0.2 mm doesn’t change the stage, but is recorded with abbreviations that indicate the type of special test used to find the spread.
If the area of cancer spread is at least 0.2 mm , but still not larger than 2 mm, it is called a micrometastasis . Micrometastases are counted only if there aren’t any larger areas of cancer spread. Areas of cancer spread larger than 2 mm are known to influence outlook and do change the N stage. These larger areas are sometimes called macrometastases, but are more often just called metastases.
NX: Nearby lymph nodes cannot be assessed .
N0: Cancer has not spread to nearby lymph nodes.
N1c: Both N1a and N1b apply.
N3: Any of the following:
N3a: either:
N3b: either:
How Is The Stage Determined
The staging system most often used for breast cancer is the American Joint Committee on Cancer TNM system. The most recent AJCC system, effective January 2018, has both clinical and pathologic staging systems for breast cancer:
- The pathologic stage is determined by examining tissue removed during an operation.
- Sometimes, if surgery is not possible right away or at all, the cancer will be given a clinical stage instead. This is based on the results of a physical exam, biopsy, and imaging tests. The clinical stage is used to help plan treatment. Sometimes, though, the cancer has spread further than the clinical stage estimates, and may not predict the patients outlook as accurately as a pathologic stage.
In both staging systems, 7 key pieces of information are used:
- The extent of the tumor : How large is the cancer? Has it grown into nearby areas?
- The spread to nearby lymph nodes : Has the cancer spread to nearby lymph nodes? If so, how many?
- The spread to distant sites : Has the cancer spread to distant organs such as the lungs or liver?
- Estrogen Receptor status: Does the cancer have the protein called an estrogen receptor?
- Progesterone Receptor status: Does the cancer have the protein called a progesterone receptor?
- HER2 status: Does the cancer make too much of a protein called HER2?
- Grade of the cancer : How much do the cancer cells look like normal cells?
In addition, Oncotype Dx® Recurrence Score results may also be considered in the stage in certain situations.
Recommended Reading: How Bad Is Breast Cancer
Comparison Of Roc Analysis Of Adc Histogram Parameters In Distinguishing Low Ki
The results of the ROC analysis of the significant ADC histogram parameters are shown in Figure 5 and Table 5. The AUC values of the mean, and the 1st, 10th, 50th, 90th, and 99th percentiles for predicting Ki-67 proliferation status for patients with T1 stage IDC were 0.816, 0.856, 0.849, 0.815, 0.755, and 0.717, respectively. Using the method developed by Delong et al., pairwise AUC value comparisons among the mean, 1st percentile, 10th percentile, 50th percentile, 90th percentile, and 99th percentile were performed. As a result, the P-value of 0.05 was adjusted to 0.0033. The 1st and 10th percentiles had relatively higher AUC values, and the two values were not significantly different . The 1st and 10th percentiles had higher AUC values than the mean or 50th percentile, but the difference was not significant . The mean and 50th percentile both demonstrated significantly higher AUC values than the 90th and 99th percentiles .
Figure 5Table 5
Ethics And Consent Statement
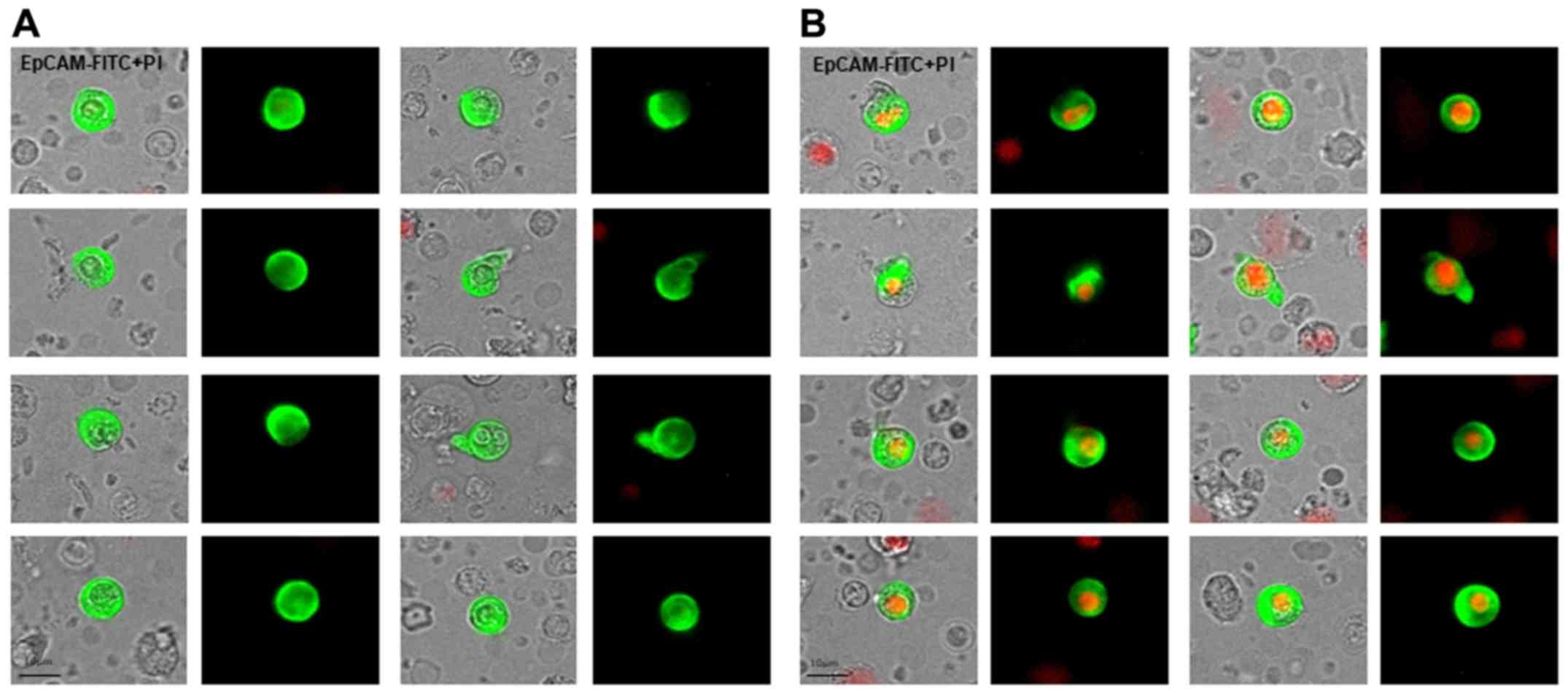
The authors declare that this is a retrospective study and it does not require approval by the Ethics Committee, but rather, only required approval by the Breast Unit Core Team Institutional Review Board . Professor Amanti, as an author of the paper, recused himself from the review and approval process undertaken by the institutional review board.
Patient consent to review their medical records was not required by the board because all patients included in the study signed, at admission, a standard form about privacy and processing of personal data.
Read Also: How To Search For Breast Cancer
Test Of Equality Of Survival Distributions For Different Levels Of Ki
Figure 2 shows that patients with Ki-67 < 15% exhibited better OS than those with Ki-67 > 15% .Figure 3 illustrates that patients with Ki-67 > 15% were more likely to develop recurrence and distant metastasis than those with Ki-67 < 15% .
Disease free survival of breast cancer patients. Patients with Ki-67 > 15% are more likely to develop recurrence and distant metastasis than those with Ki-67 < 15% .
Statistic Analysis And Software
The prospective database was built by Microsoft® Access. The statistical analysis was carried out by IBM-SPSS®. In order to compare categorical and continuous variables, Chi-square test and Students t test were used. Disease-Related Survival and Disease-Free Survival were calculated from the surgical procedure, plotting the curves by the KaplanMeier method and the Log-rank test was used for statistical comparisons.
You May Like: What Is The Rarest Form Of Breast Cancer
How Do You Read A Ki
The correct way of visual calculating Ki 67 is by counting the total number of positive-staining tumor cells in each image/field and count the total number of tumor cells in each image to calculate the Ki67 index. Calculation of the Ki67 index = No.of positive tumor cells/total No.of tumor cells ×100.
What Does It Mean If My Carcinoma Is Called Invasive Ductal Carcinoma Invasive Lobular Carcinoma Or Carcinoma With Ductal And Lobular Features
Breast carcinomas are often divided into 2 main types: invasive ductal carcinoma and invasive lobular carcinoma, based on how they look under the microscope. In some cases, the tumor can have features of both and is called a mixed ductal and lobular carcinoma. Another term for invasive ductal carcinoma is invasive mammary carcinoma of no special type, because it is the most common type of breast carcinoma.
Both invasive ductal carcinomas and invasive lobular carcinomas arise from the cells lining the ducts and lobules in the breast. In general, invasive lobular and invasive ductal carcinomas of the breast arent treated differently.
Recommended Reading: Can You Feel If You Have Breast Cancer
What If My Report Mentions Her2/neu Or Her2
Some breast cancers have too much of a growth-promoting protein called HER2/neu . The HER2/neu gene instructs the cells to make this protein. Tumors with increased levels of HER2/neu are referred to as HER2-positive.
The cells in HER2-positive breast cancers have too many copies of the HER2/neu gene, resulting in greater than normal amounts of the HER2 protein. These cancers tend to grow and spread more quickly than other breast cancers.
All newly diagnosed breast cancers should be tested for HER2, because women with HER2-positive cancers are much more likely to benefit from treatment with drugs that target the HER2 protein, such as trastuzumab , lapatinib , pertuzumab , and T-DM1 .
Testing of the biopsy or surgery sample is usually done in 1 of 2 ways:
- Immunohistochemistry : In this test, special antibodies that will stick to the HER2 protein are applied to the sample, which cause cells to change color if many copies are present. This color change can be seen under a microscope. The test results are reported as 0, 1+, 2+, or 3+.
- Fluorescent in situ hybridization : This test uses fluorescent pieces of DNA that specifically stick to copies of the HER2/neu gene in cells, which can then be counted under a special microscope.
Many breast cancer specialists think that the FISH test is more accurate than IHC. However, it is more expensive and takes longer to get the results. Often the IHC test is used first:
Original Articledo You Know The Ki
Limiting the number of observers can improve reproducibility of Ki-67 assessments.
-
Comparison with published data-sets helps to define adequate Ki-67 cut-off levels.
-
Lobular breast cancers have a significantly lower Ki-67 than ductal breast cancers.
-
Ki-67 values and standard deviation increase parallel to increasing tumor grade.
-
Ki-67, properly assessed, remains important for decision making in breast cancer.
Read Also: How To Know If Breast Cancer Spread
What Does It Mean If My Doctor Asks For A Special Molecular Test To Be Performed On My Specimen
Molecular tests such as Oncotype DX® and MammaPrint® may help predict the prognosis of certain breast cancers, but not all cases need these tests. If one of these tests is done, the results should be discussed with your treating doctor. The results will not affect your diagnosis, but they might affect your treatment.
Ki67 In Neoadjuvant Endocrine Therapy
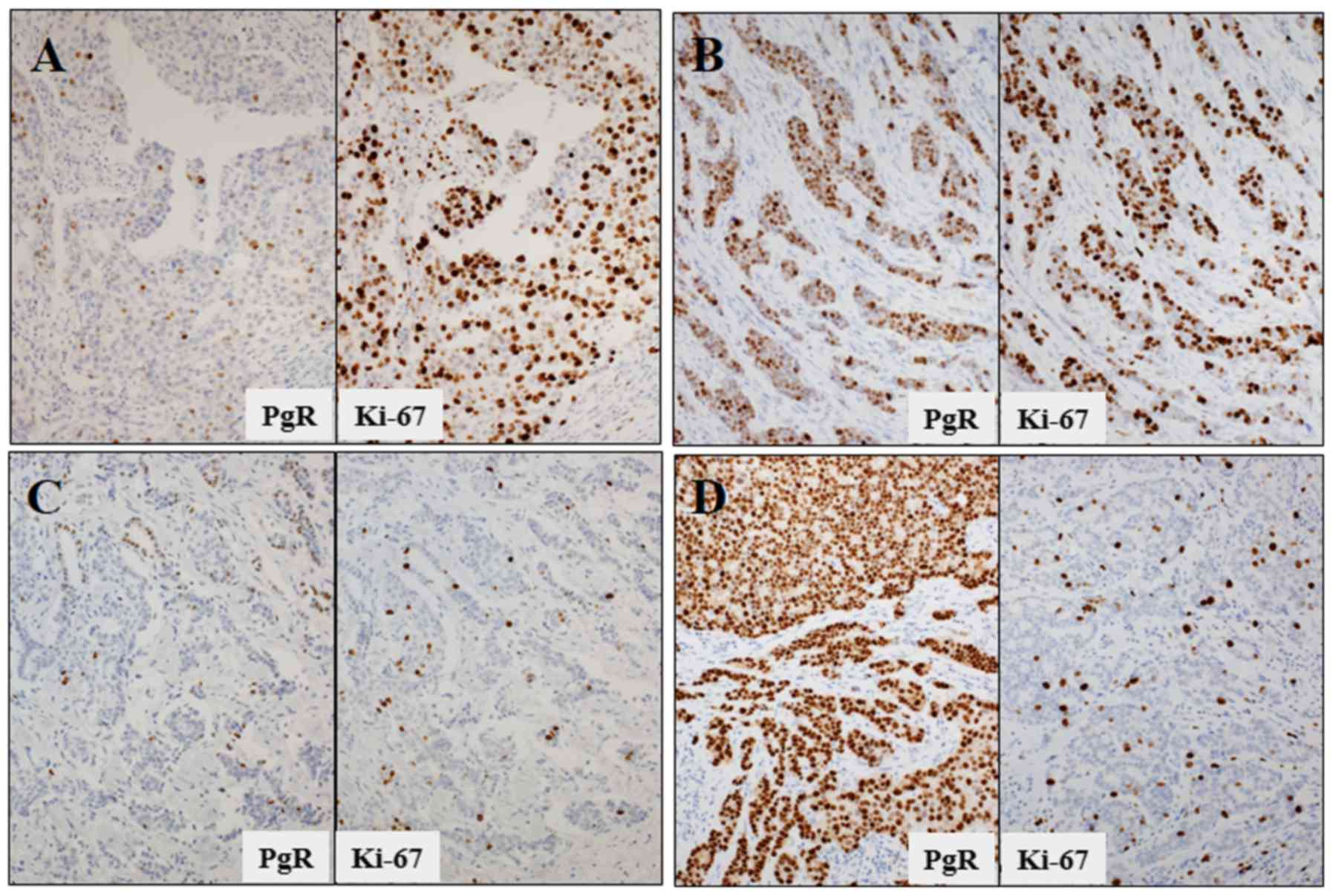
Table 1 Main neoadjuvant endocrine trials.
P024 was a randomized, multinational, double-blind study comparing 4 months of letrozole vs. tamoxifen in postmenopausal women with hormone-responsive primary untreated breast cancer . P024 indicated that the percentage of Ki67-positive cells, pathological tumor size, lymph node status, and ER status were independently associated with breast cancer-specific survival and relapse-free survival. Based on these factors, Ellis et al. obtained a clinically valuable prognostic model of preoperative endocrine prognostic index score for the outcome prediction of hormone-positive breast cancer with NET. The Ki67 and PEPI triage approaches can predict the risk of relapse. NET was initially an option for breast cancer patients who were too frail to have surgery or cytotoxic chemotherapy. It is very difficult to evaluate the efficacy of adjuvant endocrine therapy because of its long-term follow-up, and NET offers useful clues. The initial endocrine neoadjuvant therapy clinical trial collected data to evaluate the long-term outcome of adjuvant endocrine therapy indirectly rather than as a neoadjuvant treatment . Future adjuvant endocrine therapy clinical research designs should be based on a biological superiority hypothesis generated by a neoadjuvant endocrine study .
Also Check: How To Do An At Home Breast Cancer Check
What Is Considered A High Ki
What is considered a high Ki-67 score in breast cancer? Understanding Your Ki-67 Results
Less than 10% is considered low. 20% or higher is considered high.
Does a high Ki-67 mean chemo? In subgroup analysis, a higher Ki-67 value was a significant predictive factor for the response to neoadjuvant chemotherapy, especially in ER-negative and HER2-positive breast cancer patients.
What is normal Ki-67? A Ki-67 index of 2% or lower means that fewer than 2 in every 100 cells are dividing. This is a grade 1 NET . This means that between 3 and 20 cells in every 100 cells are dividing.
What is Ki-67 a marker for? The Ki67 protein is well characterized at the molecular level and is extensively used as a prognostic and predictive marker for cancer diagnosis and treatment. Increasing evidence indicates that Ki67 may be an effective target in cancer therapy.
Understanding Your Pathology Report: Breast Cancer
When your breast was biopsied, the samples taken were studied under the microscope by a specialized doctor with many years of training called a pathologist. The pathologist sends your doctor a report that gives a diagnosis for each sample taken. Information in this report will be used to help manage your care. The questions and answers that follow are meant to help you understand medical language you might find in the pathology report from a breast biopsy, such as a needle biopsy or an excision biopsy.
In a needle biopsy, a needle is used to remove a sample of an abnormal area. An excision biopsy removes the entire abnormal area, often with some of the surrounding normal tissue. An excision biopsy is much like a type of breast-conserving surgery called a lumpectomy.
Recommended Reading: What Is Er+ Breast Cancer
How Do Doctors Use Ki67
Clinicians have made wide use of Ki-67 staining with immunohistochemistry as a prognostic biomarker in breast cancer for years.1 The International Ki-67 in Breast Cancer Working Group is a panel of breast cancer experts that has confirmed the evidence for Ki-67s clinical value as a prognostic biomarker and clinical studies support Ki-67 measurement to help guide treatment decisions.2-5
Establishment Of The International Ki67 In Breast Cancer Working Group
In 2011, we established the International Ki67 in Breast Cancer Working Group to review methods of determination of Ki67 levels in breast cancer . Because of the multiplicity of assays and the apparent poor standardization of them for this marker, we set out to establish internationally acceptable methods for the determination of Ki67. Since 2011, as described in the remainder of this article, we and others have made substantial efforts to address both the technical and scoring aspects of Ki67 assessment. Nonetheless, current guidelines remain skeptical about the technical validity of Ki67 IHC assays. For example, the recent American Joint Committee on Cancer guideline on cancer staging states As a single factor, Ki-67 was not considered a reliable factor for implementation in clinical practice because of the known lack of reproducibility . Consistent or similar statements are present in many national and international guideline documents .
Also Check: Is It Possible To Have Breast Cancer At 14
M Categories For Breast Cancer
M followed by a 0 or 1 indicates whether the cancer has spread to distant organs — for example, the lungs, liver, or bones.
M0: No distant spread is found on x-rays or by physical exam.
cM0: Small numbers of cancer cells are found in blood or bone marrow , or tiny areas of cancer spread are found in lymph nodes away from the underarm, collarbone, or internal mammary areas.
M1: Cancer has spread to distant organs as seen on imaging tests or by physical exam, and/or a biopsy of one of these areas proves cancer has spread and is larger than 0.2mm.
Histopathological Review And Tissue Microarray Construction
Details of centralised pathology review have been previously described. Documented pathological features included histological type, histological grade,, and presence of lymphovascular invasion by breast cancer . Viable tumour samples from different areas of paraffin blocks were selected for inclusion in tissue microarrays with three 0.6mm cores sampled from each case using a manual arrayer . Slides, prepared from 4µm thick sections, were stained with haematoxylin and eosin together with an immunohistochemical panel and human epidermal growth factor 2 silver in situ hybridisation .
Read Also: How To Use Cannabis Oil For Breast Cancer
Approval Based On Monarche Results
Verzenios approval to treat certain early-stage breast cancers after surgery is based on results from the monarchE study.
The study found that adding Verzenio to hormonal therapy after surgery to remove early-stage hormone-receptor-positive HER2-negative breast cancer with a high risk of recurrence lowers recurrence risk by about 25% versus using hormonal therapy alone. Specifically, adding Verzenio to hormonal therapy lowered the risk of distant recurrence the cancer coming back in a part of the body away from the breast, such as the bones or liver by about 28%.
The design and results of the monarchE study are practice-changing and represent the first advancement in adjuvant treatment of hormone-receptor-positive HER2-negative breast cancer in a very long time, Sara Tolaney, M.D., MPH, of the Dana-Farber Cancer Institute and monarchE investigator, said in a statement. This FDA approval for Verzenio in combination with endocrine therapy in the early breast cancer setting has the potential to become a new standard of care for this population. We are encouraged by the marked reduction in the risk of recurrence even beyond the 2-year treatment period in these patients, and I’m grateful to be able to offer this as a treatment option to my patients.
Tumor Biomarker Application In The Clinic: Important Semantics

In 2009, Teutsch et al. , representing the Evaluation of Genomic Application in Practice and Prevention initiative, described 3 critical elements to determine if a germ-line genetic test should be used to manage care: 1) analytical validity, 2) clinical validity, and 3) clinical utility. Building on the EGAPP initiative, the United States Food and Drug Administration and the National Institutes of Health convened a group of stakeholders to jointly develop a glossary to better define and harmonize biomarker terminology: the Biomarkers, Endpoints, and Other Tools Resource . Broadly, the Biomarkers, Endpoints, and Other Tools Resource defines the acceptability of analytical validity in terms of its sensitivity, specificity, accuracy, precision, and other relevant performance characteristics. The level of acceptability of analytical performance of Ki67 differs among applications and intended uses.
The efforts of the IKWG over the last decade aimed first to determine analytical validity for Ki67 IHC and to promote standardization. The IKWG has not yet formally addressed clinical utility. At the October 2019 meeting, the IKWG addressed several questions that were considered essential in order for Ki67 to be used to manage patient care.
Read Also: Is Estrogen Cream Safe For Breast Cancer Survivors